Data Integrity
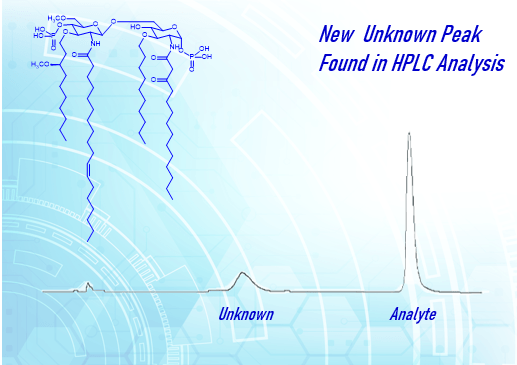
In recent years, problems with data integrity have become a key concern for global regulatory agencies. In 2015, the UK Medicines and Healthcare products Regulatory Agency (MHRA) issued a
Guidance for Industry on Data Integrity. In 2016, FDA issued a
Data Integrity and Compliance With Drug CGMP Questions and Answers Guidance for Industry. The FDA wrote, “The guidance has been developed in response to an increase in findings of data integrity lapses in recent inspections. FDA expects that all data be reliable and accurate. CGMP regulations and guidance allow for flexible and risk-based strategies to prevent and detect data integrity issues.” With this problem area identified and an area of focus for regulatory agencies, it is wise for firms to ensure CGMP compliance in this critical area.

Interested in our services? We’re here to help!
USEFUL LINKS
All Rights Reserved | Site by Fix8